名前を赤で表記しているメンバーは当研究室の学生・院生、青で表記しているメンバーは当研究室の教員です。
原著論文
- Kunihiro Narita, Yohei Ishida, Shuichi Nukui, Zhong Huang, Tetsu Yonezawa
“Surface Menshutkin SN2 Reaction on Basic Gold Clusters Provides Novel Opportunities for the Cationization and Functionalization of Molecular Metal Clusters”
The Journal of Physical Chemistry Letters, 12(49), 11761-11765 (2021).
DOI: 10.1021/acs.jpclett.1c03498 (Published (web) 2 Dec 2021)
【国際共同研究】【科研費】
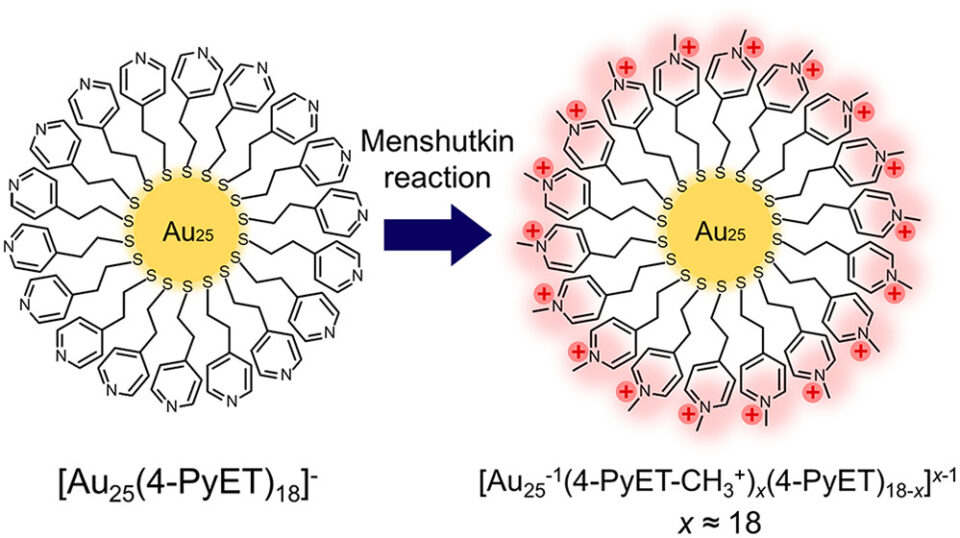
Abstract: Surface chemical reactions on atomically precise metal clusters have considerable attention for opening a new platform for cluster functionalization. In this study, basic Au25(4-PyET)18 (4-PyET = −SCH2CH2Py; Py = pyridyl) clusters were successfully transformed into cationized Au25(4-PyET-CH3+)x(4-PyET)18-x clusters, without altering their Au25 cores, through the Menshutkin SN2 reaction of their surface Py moieties. This study offers not only a simple cationization method but also a protocol for modifying the surface functionalities of molecular metal clusters via a synthetic reaction.
Cover

- Mai Thanh Nguyen, Lianlian Deng, and Tetsu Yonezawa 【Review paper】
“Control of nanoparticles synthesized via vacuum sputter deposition onto liquid: a review”
Soft Matter, 18(1), 19-47 (2022).
DOI: 10.1039/D1SM01002F (Published (web) 23 November 2021)
【研究室単独研究】【科研費】

Abstract: Sputter deposition onto a low volatile liquid matrix is a recently developed green synthesis method of metal/metal oxide nanoparticles (NPs). In this review, we introduce the synthesis method and highlight its unique features emerging from the combination of the sputter deposition and capability of regulating particle growth by the liquid matrix. Then, manipulating the synthesis parameters to control particle size, composition, morphology, and crystal structure of NPs is presented. Subsequently, we evaluate the key experimental factors governing the particle characteristics and the formation of monometallic and alloy NPs to provide overall directions and insights into the preparation of NPs with desired properties. Following that, the current understanding of the growth and formation mechanism of sputtered particles in liquid media, in particular, ionic liquids and liquid polymer, during and after sputtering is emphasized. Finally, we discuss the remaining challenges and share our perspective in the future prospect of the synthesis method and the obtained NPs.
- Mohan Gopalakrishnan, Ahmad Azmin Mohamad, Mai Thanh Nguyen, Tetsu Yonezawa, Jiaqian Qin, Patchanita Thamyongkit, Anongnat Somwangthanaroj, and Soorathep Kheawhom
“Recent advances in oxygen electrocatalysts based on tunable structural polymers”
Materials Today Chemistry, 23, 100632 (2022) (Mar 2022) 【Open Access】
DOI: 10.1016/j.mtchem.2021.100632 (Published (web) 19 November 2021)
【国際共同研究】【JST】
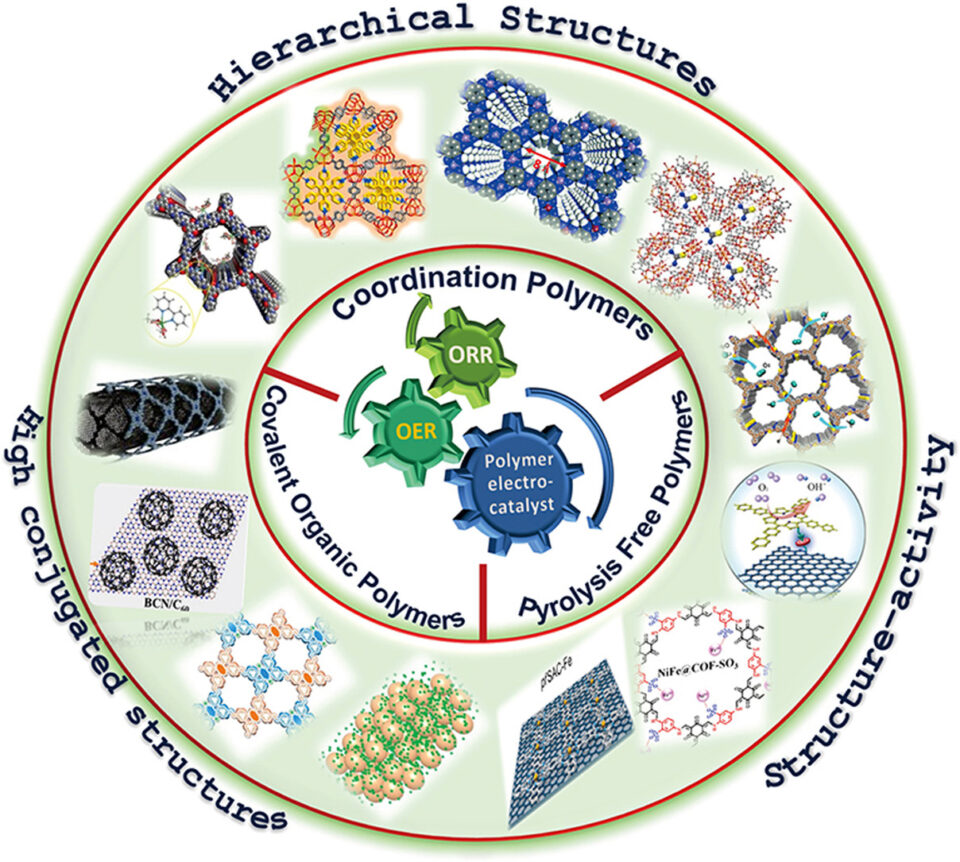
Abstract: Due to their high energy density, great safety and eco-friendliness, zinc-air batteries (ZABs) attract much attention. During the process of charging and discharging, the two key processes viz. oxygen evolution reaction (OER) and oxygen reduction reaction (ORR) limit their efficiency. In general, the noble metal-based electrocatalysts (ORR: platinum (Pt); OER: iridium (IV) oxide [IrO2] and ruthenium oxide [RuO2]) have long been used. Nonetheless, these noble metal electrocatalysts also have their limitations owing to high cost and poor stability. As alternatives, polymers are found to be most promising on account of their tunable structure, uniform network, high surface morphology and strong durability. Polymers are capable catalysts. In this review, recent advances as well as insight into the architecture of covalent organic polymers (COPs), metal coordination polymers (MCPs) and pyrolysis-free polymers (PFPs) are duly outlined.
- Mohammad Etesami, Ahmad Azmin Mohamad, Mai Thanh Nguyen, Tetsu Yonezawa, Rojana Pornprasertsuk, Anongnat Somwangthanaroj, and Soorathep Kheawhom
“Benchmarking superfast electrodeposited bimetallic (Ni, Fe, Co, and Cu) hydroxides for oxygen evolution reaction”
Journal of Alloys and Compounds, 889, 161738 (2022).
DOI: 10.1016/j.jallcom.2021.161738 (Published (web) 28 Aug 2021)
【国際共同研究】【JST】Abstract: High-performing electrocatalysts of oxygen evolution reaction (OER) are essential for various clean energy devices. Due to their excellent OER performance, transition metal layered doubled hydroxides (LDHs) attract increasing interest. Herein, a systematic study of superfast single-step synthesis for a series of carbon-free, binder-free self-supported bimetallic hydroxides on Ni foam (NF) and their associated performance towards OER in alkaline electrolytes is presented. Transition metal hydroxides are directly deposited on NF by a superfast electrodeposition technique. The resultant bimetallic hydroxides are uniformly distributed over NF in short-range order crystalline or amorphous structures. All six electrodes demonstrate significantly greater OER activities than ruthenium oxide (RuO2). The interaction between transition metal LDHs and NF determines OER performance. Applying optimum conditions, the highest activities are accomplished. Overall, results show excellent OER performance. The fast electrodeposition technique provides a systematic and cost-effective way to achieve durable self-supported transition metal LDH structures showing excellent OER performance.
- Natta Jaikrajang, Wathanyu Kao-Ian, Tatsuki Muramatsu, Rungroj Chanajaree, Tetsu Yonezawa, Zakaria Y. Al Balushi, Soorathep Kheawhom, and Rongrong Cheacharoen
“Impact of Binder Functional Groups on Controlling Chemical Reactions to Improve Stability of Rechargeable Zinc-Ion Batteries”
ACS Applied Energy Materials, 4(7), 7138-7147 (2021).
DOI: 10.1021/acsaem.1c01214 (Puublished (web) 30 June 2021)
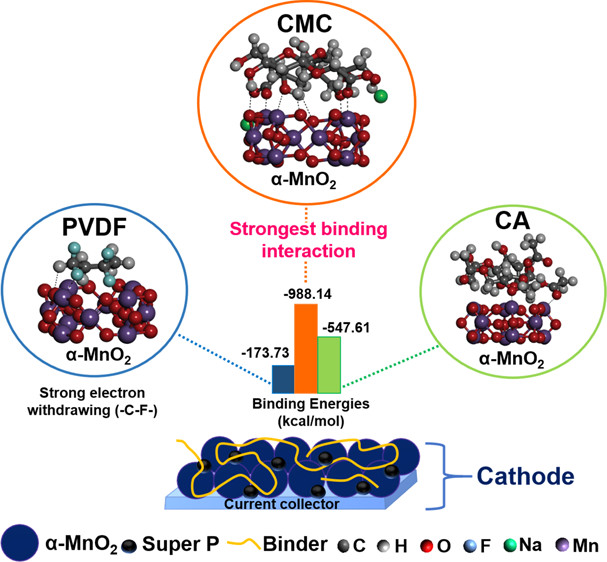
Abstract: Rechargeable zinc-ion batteries (ZIBs) are promising alternatives for large-scale energy storage systems. However, instability of the cathode during operation leads to rapid capacity fading and poor stability. Binders play a crucial role in keeping all components in the cathode intact during cycling. However, the impact of the binders’ chemical structure on the electrochemical reaction in ZIBs is not well understood. Herein, the effect of the chemical structure of a conventional polyvinylidene fluoride (PVDF) and green binders, that is, sodium carboxymethyl cellulose (CMC) and cellulose acetate (CA), on the performance, cyclability, and reaction of Zn/α-MnO2 aqueous batteries is investigated. Results show that a cathode having a PVDF binder yields the highest specific capacity in a full battery. Besides, the CMC-based ZIB is seen to attain superior cycling stability through 500 galvanostatic charge–discharge (GCD) cycles having no irreversible products confirmed by scanning electron microscopy (SEM) and X-ray diffraction (XRD). It is found that the Na+ ion in the CMC structure plays a critical role in promoting prominent battery reactions. The CMC-based ZIB, therefore, is able to maintain its high ionic diffusivity obtained from the galvanostatic intermittent titration technique (GITT) during prolonged operation. Moreover, the interaction between binders and α-MnO2 has been investigated via density-functional theory (DFT) to affirm the high stability of the ZIB/α-MnO2 with the CMC binder. This work highlights the importance of the selection of functional groups on the binder not only to enhance stability but also to control the preferential reactions of batteries. Such findings are ultimate keys for the development of low-cost, stable, and eco-friendly energy storage devices.
- Nikdalila Radenahmad, Ramin Khezri, Ahmad Azmin Mohamad, Mai Thanh Nguyen, Tetsu Yonezawa, Anongnat Somwangthanaroj, Soorathep Kheawhom
“A durable rechargeable zinc-air battery via self-supported MnOx-S air electrode”
Journal of Alloys and Compounds, 883, 160935 (11 pages) (2021). 【Open Access】
DOI: 10.1016/j.jallcom.2021.160935 (Published (web) 23 June 2021)Abstract: The durability and performance of an air electrode can have a crucial impact on rechargeable zinc-air batteries (ZABs). A typical air electrode fabricated using conductive carbon and a polymeric binder suffers rapid degradation during charging, stemming from oxygen bubbles erosion and carbon corrosion. This work presents a durable air electrode having a self-supported sulfur-doped manganese oxides (MnOx-S) electrocatalyst prepared through hydrothermal process followed by ambient temperature sulfurization. Upon sulfurization,
MnO2 nanoflakes are transformed into MnOx-S having a heterostructure of Mn3O4, MnO2 and MnSx. MnOx-S provided superior electrochemical properties for both oxygen reduction reaction (Tafel slope of 68 mV/dec) and oxygen evolution reaction (Tafel slope of 80 mV/dec). Self-supported electrodes using MnOx-S and MnO2 electrocatalysts were scrutinized in a bi-electrode rechargeable ZAB having a stagnant electrolyte. In the ZAB using the self-supported MnOx-S electrode, both significant discharge peak power density (74 mW/cm2 at 135 mA/cm2) as well as low voltage difference between charge and discharge processes (0.75 V) were observed. In addition, for charge-discharge cycling at 20 mA/cm2, the ZAB using the self-supported MnOx-S electrode achieved stable cycling through 2000 cycles without apparent degradation. The air electrode having the self-supported MnOx-S paves the way for a more durable and higher performance favoring practical application for rechargeable ZABs. - Shilei Zhu, Mai Thanh Nguyen, and Tetsu Yonezawa* (Review Paper)
“Micro and Nano Encapsulated Metal and Alloy Based Phase Change Materials for Thermal Energy Storage”
Nanoscale Advances, 3(16), 4626-4645 (2021).【Open Access】
DOI: 10.1039/D0NA01008A (Published (web) 14 June 2021)

Abstract: An overview of recent literature on the micro- and nano-encapsulation of metallic phase-change materials (PCMs) is presented in this review to facilitate an understanding of the basic knowledge, selection criteria, and classification of commonly used PCMs for thermal energy storage (TES). Metals and alloys with high thermal conductivity can be used as PCMs for rapid heat storage in compact systems owing to their high volumetric TES density. The emerging application of metal PCMs in different fields such as solar thermal energy management, smart wearable devices with thermal comfort control, and cooling of electronic devices call for the need of micro- and nano-TES particles, which can be synthesised in different forms to satisfy specific requirements. As metals are easily oxidised, especially at the micro- and nano-level, encapsulation of metal-based PCM particles is important for sustainable use at high operating temperature in ambient conditions. Recent studies focusing on the encapsulation of metallic PCMs at the micro- and nano-level have been reviewed and classified in terms of the melting point of metal/alloy PCMs used and types of encapsulation materials, such as oxides, polymers, carbon, and metals. The current review is expected to provide an outlook on novel metal and alloy PCMs with function-directed structures and superior TES properties for a broad range of applications.
Selected as a front page.

- Mingbei Zhu, Mai Thanh Nguyen, Yuen-ting Rachel Chau, Lianlian Deng, and Tetsu Yonezawa
”Pt/Ag Solid Solution Alloy Nanoparticles in Miscibility Gap Synthesized by Co-Sputtering onto Liquid Polymer”
Langmuir, 37(19), 6096-6105 (2021).
DOI: 10.1021/acs.langmuir.1c00916 (Published (web) 7 May 2021)

Abstract: Pt/Ag solid solution alloy nanoparticles (NPs) with mean size below 3 nm were obtained with composition in miscibility gaps by cosputtering onto liquid polyethylene glycol (PEG, MW = 600). Adjusting the sputtering currents from 10 to 50 mA did not influence the particle sizes obviously but caused a substantial difference in the composition and distributions of Pt/Ag NPs. This is different from sputtered Pt/Au NPs where particle size is correlated with composition. For a pair of sputtering currents, the formed Pt/Ag alloy NPs have a range of compositions. The normal distribution with Pt of 60.2 ± 16.2 at % is observed for the Pt/Ag sample with a nominal Pt content of 55.9 at %, whereas Pt-rich (85.1 ± 14.0 at % Pt) and Ag-rich (19.8 ± 12.2 at % Pt) Pt/Ag samples with nominal Pt contents of 90.9 and 11.9 at % contain more pure Pt and pure Ag NPs, respectively. Different from NPs obtained in PEG, the sputtered NPs on TEM grids had more uniform composition for a longer sputtering time along with a significant increase of particle sizes. This reveals that PEG hindered the combination of NPs and clusters, resulting in small particle sizes even for long time sputtering and broader composition distributions. Thus, the samples obtained in PEG have the compositions mainly determined by the random atom combination in the vacuum chamber and possibly in initial landing of atom/clusters on the PEG surface.
- Nikdalila Radenahmad, Ramin Khezri, Ahmad Azmin Mohamad, Mai Thanh Nguyen, Tetsu Yonezawa, Anongnat Somwangthanaroj, and Soorathep Kheawhom
“Highly Stable Rechargeable Zinc-ion Battery using Dimethyl Sulfoxide Electrolyte”
Materials Today Energy, 21, 100738 (2021). 【Open Access】
DOI: 10.1016/j.mtener.2021.100738 (Published (web) 2 April 2021).Abstract: Due to their high safety, low-cost, eco-friendliness and impressive electrochemical performance, rechargeable zinc-ion batteries (ZIBs) show great potential as electrical energy storage devices for large-scale application. Nonetheless, recently developed ZIBs still suffer from low cycling stability and high capacity fading. Such shortcomings are caused by the reversibility of both zinc (Zn) and the cathode host material as well as hydrogen evolution in aqueous electrolytes, which are naturally protic solvent. Herein, dimethyl sulfoxide (DMSO), a polar aprotic solvent, is examined as an electrolyte for a ZIB. Zn stripping/plating in DMSO-based electrolytes shows excellent reversibility and dendrite-free morphology. During charging and resting modes, hydrogen evolution is effectively inhibited. Insertion/extraction of Zn ions in DMSO-based electrolytes into delta-type manganese dioxide (δ-MnO2) demonstrates high stability, achieving a decent initial capacity of 159 mAh/g at 50 mA/g and a nominal discharge voltage of 1.15 V. At 100 mA/g charge/discharge cycling, the ZIB, having the DMSO-based electrolyte, can pass 1000 cycles, displaying a capacity retention of 60%. Overall, improved performance of ZIBs can be attained using DMSO-based electrolytes. Results pave the way towards the practical application of ZIBs.
- Ikumi Akita, Yohei Ishida, and Tetsu Yonezawa
“Direct imaging of individual organic molecules in supramolecular assembly strongly fixed via multivalent electrostatic interactions”
The Journal of Physical Chemistry C, 125(8), 4917-4923 (2021).
DOI: 10.1021/acs.jpcc.1c00738 (Published (web) 23 Feb 2021)

Abstract: Recent developments in electron microscopy and novel techniques for the precise control of low-dimensional substrate materials have led to the emergence of direct imaging of organic molecules. However, supramolecular assemblies constructed via noncovalent interactions are structurally unstable under electron beam irradiation. This study focused on a supramolecular assembly of well-isolated organic molecules based on multivalent electrostatic interactions, where anionically charged clay mineral nanosheets were used as a host material. Direct imaging of the single organic molecules and supramolecular assemblies on the monolayer clay mineral nanosheets was achieved via annular dark field scanning transmission electron microscopy using heavy metal atom markers. The versatility of multivalent electrostatic interactions for the stable fixation of organic molecules during electron microscopic imaging was demonstrated. This strategy can be applied to a wide range of supramolecular systems comprising various guest molecules and host materials, thereby broadening the scope of atomic-scale imaging.
総説・解説
- 米澤 徹
「導電性・熱伝導性ペーストに向けた金属微粒子・ナノ粒子」
粉体技術, 13(12), 19-26 (2021). - Tetsu Yonezawa, Shilei Zhu, and Mai Thanh Nguyen
Chapter 15 “Miscellancous Reductants” in
“Reducing Agents in Colloidal Nanoparticle Synthesis” pp.393-459.
Edited by Stefanos Mourdikoudis (University College London, UK)
Nanoscience and Nanotechnology Series 50.
The Royal Society of Chemistry (2021)
ISBN 978-1-83916-165-0

- 米澤 徹
「銅微粒子を用いた低温焼成導電材料の構築」
ナノ学会会報, 19(2), 57-61 (2021).
